4th semester

DC1 ALESSANDRO CUZZERI OCTOBER 24 - MARCH 25
Months 19-21 (October - December 2024): Participation in the Tawani 2025 Lake Untersee Expedition, Antarctica (Oct – Dec 2024).
November 2024: Tawani 2025 Lake Untersee Expedition, Antarctica. Sampling and preliminary sample preparation procedures. Biostatistical analyses for own paper (Intra-seasonal trends of cryoconite bacterial communities on an Alpine Glacier).
December 2024: Tawani 2025 Lake Untersee Expedition, Antarctica. Sampling and preliminary sample preparation procedures. Biostatistic analyses for own paper (Intra-seasonal trends of cryoconite bacterial communities on an Alpine Glacier).
Month 22 (January 2025): Data analysis planning for Antarctica samples in collaboration with Team Anesio and drafting of policy brief on “Geotextiles on Alpine glaciers: science-driven recommendations and good practices”.
February 2025)– Total-RNA pipeline setup for upcoming analyses on local HPC cluster. Biostatistical analyses and beginning of drafting of own paper (Intra-seasonal trends of cryoconite bacterial communities on an Alpine Glacier).
Month 24 (March 2025) – Drafting of deliverable “An estimation of the microbial biomass and activity originating from the atmosphere and deposited on glacier surfaces”. Sequencing and bioinformatics of samples from 2023 Antarctica expedition..

DC6 KLARA KOHLER NOVEMBER 24 - APRIL 25
At the beginning of November, I attended the Nordic Branch Meeting, an annual conference of the Scandinavian branch of the International Glaciological Society (IGS). In mid-November, I had my midterm evaluation, which included a 45- minute presentation about my work and the status of my PhD, followed by approximately one hour of questions from my external examiner, Professor Kate Hendry (Ocean Climate Scientist at the British Antarctic Survey).
In December, I carried out the first round of a crushing experiments with sediment samples from Ilulissat, Greenland, and measured the difference in concentration of nutrients (N, P, Si, Fe) released into solution from the crushed and non- crushed sediments. At the end of December, I visited the GFZ in Potsdam, Germany, for a week to carry out XRD measurements on these crushed and uncrushed sediment samples.
At the beginning of January, I attended the Greenland Ice Sheet Seminar at our Risø Campus in Roskilde, Denmark. For the rest of the month I concentrated on organising and preparing the lab experiments that I started at the end of January.
In February, I spent most of the days in the lab. I started a time series on the nutrient dynamics between glacier sediments and pore water, measuring how the concentrations of N, P, Si and Fe change over time between dissolved in the pore water and available at the sediment surface. This time series will last about six months. In mid-February, I also attended the PhD Bundle Meeting, a meeting for all PhD students and supervisors in our department; this year's topic was “Insights and Reflections on the PhD Experience.”
In March I continued lab work. I continued working on the time series and conducted a second crushing experiment, where I crushed sediments and measured the concentration of released nutrients from the sediment into solution. For this experiment I used glacial sediments that I collected in Kangerlussuaq, Greenland in the summer of 2024. I also took care of the preparations for my fieldwork in September and October this year, for which I had to pack and ship all the equipment I need for the expedition.
In April, I started processing the data from this year's lab work. At the end of April, I attended the EGU Conference in Vienna, Austria, together with other PhD students from our IceBio network. At the conference, I gave a talk about my research topic, glacial flour, and how it serves as a nutrient source for the subglacial and downstream environment.

DC7 MARCO AJMAR JAN 25 - June 25
Month 18 (January 2025): I started writing my PhD transfer report and prepared sampling gears for Svalbard fieldwork.
Month 19 (February 2025): I measured dissolved silica, phosphate and alkalinity of Iceland 2024 river water samples. I also carried out ICP-OES analyses of the same river samples plus snow and ice samples from Svalbard 2023 fieldwork. I analysed and processed all data gathered during lab-work.
Month 20 (March 2025): I finished writing my PhD transfer report and prepared its presentation. I also set up a suitable power source for nutrient sensor deployment. I then tested phosphate and nitrate sensors in a lake in Potsdam and started packing for the Iceland fieldwork in April.
Month 21 (April 2025): After presenting my PhD transfer report, I completed fieldwork packing and preparation. From 09/04 to 17/04 I was on secondment/fieldwork in Iceland near the Langjokull glacier. We deployed for five days in a glacial river (Hvita) nitrate, phosphate and dissolved silica sensors from ClearWater Sensors, alongside pH, turbidity, conductivity, dissolved oxygen and dissolved organic matter sensors. We also sampled the glacial river from the glacier to the fjord for both inorganic and organic dissolved nutrients.
Month 22 (May 2025): I conducted a three week long secondment at Aarhus University for low level dissolved P and N species analysis on samples from Svalbard 2025 and Greenland 2024 fieldworks. I did DNA extractions (together with DC5 Anirban Majumder) of samples from Iceland 2024 and Svalbard 2025 fieldwork.
Month 23 (June 2025) I was on secondment (with DC6 Klara Koehler) at ClearWater Sensors in Southampton for sensors testing, maintenance and deployment planning. We processed data obtained during the secondment at Aarhus University and started packing for the August Summer fieldwork. I also completed a draft of the nutrient sensors deliverable

DC8 SILJE WAALER FEBruary 25 - JULY 25
Month 19 (Feb. 2025): I went to the British Antarctic Survey in Cambridge to undertake the analysis of DSi collected in Svalbard and mainland Norway as well as sequential extraction on sediments from Kongsfjorden. This involved learning how to operate a SEAL AA500 Segmented Flow Analyser and adjust to accommodate matrix of seawater.
Month 20 (March 2025): I focused on analysing data retrieved through my visit at BAS and NTNU. Moreover, I looked into the CTD data retrieved from our field campaigns in Kongsfjorden using a RStudio script provided by NPI researcher Allison Bailey.
Month 21 (April 2025): The work conducted in March continued into April. Time was also spent on planning out my upcoming secondment at AU in May.
Month 22 (May 2025): I made a thorough plan for my secondment at AU and read up on literature related to the topic. Half way into May went to AU and started extracting DNA from my samples. Moreover, I also did amplicon sequencing for 16s in collaboration Assistant Prof. Thanassis Zervas.
Month 23 (June 2025): I continued the work on microbial molecular techniques as part of my secondment at AU. Moreover, I started doing qPCR on target genes involved with nitrogen cycling. During my secondment at AU, I also started writing on my introduction and method section for my first paper.
Month 24 (July 2025): I went back to AU despite my secondment being over, to conduct my dissolution experiment at AU in collaboration with ICEBIO DC Klara Koehler and Postdoc. Beatriz Gill Olivas. In this period, I also worked on my deliverable for ICEBIO on glacial flours ability to increase crop yield.

DC3 ANNIKA MORISCHE february 25 - July 25
Month 19 (Feb. 25) This month, my work was focused on participation in a sampling campaign to the Greenlandic Sea, aimed at sample collection for studying winter carbon dynamics. Particular attention was given to capturing potential subglacial carbon signatures, which are most detectable during winter when surface meltwater input is minimal, thus reducing signal dilution.
Month 20: March 2025 was dedicated to debriefing the fieldwork and holding a meeting with collaboration partners for analysis of collected water samples for DNA/RNA in addition to chemical characterization efforts. Moreover, after initial improvement of extraction strategy and fine-tuning of analysis parameters, first results from 13C incubations collected in Greenland 2024 showed successful isotope enrichment, prompting scientific discussions to further improve compound detection. Based on these findings, a methodological adjustment was proposed, shifting from GC-MS to LC-MS analyses to better resolve polar metabolites.
Month 21: Results from stress response analysis in glacial communities were prepared for and presentation at the EGU conference in Vienna. The feedback gathered during this conference was directly integrated into the ongoing statistical analysis of metabolic shifts in response to changing abiotic stressors.
Month 22: For securing 13C enrichment results to resolve metabolites produced by the autotrophic community on glacier surfaces, moth 3 was focused on planning the next field campaign to Greenland. Discussions were also initiated about expanding sampling efforts for mass spectrometry-based collaborations, targeting characterization of glacial carbon pools and natural 13C abundance across different glacial habitats. Work on the first manuscript was intensified, centered on the stress response analysis.
Month 23: Fieldwork preparations entered their final phase in June 2025, with plans to sample two timepoints and increase replication in order to enable metabolite flux analysis as successful enrichment has been confirmed by sample analysis from the previous year. This included the selection of sampling locations, detailed logistical planning, and the preparation of carbon-clean labware and other essential field equipment. In parallel, discussions were held regarding analyses for the third planned paper, including sequencing of samples collected during the earlier Greenlandic Sea campaign.
Month 24: July 2025 was marked by a return to the field, with intensive work carried out in Kangerlussuaq, Greenland. Activities included conducting 13C surface ice incubations, performing PAM (Pulse-Amplitude-Modulated fluorometry) measurements, and collecting samples for both mass spectrometry characterization and sequencing. Additionally, in collaboration with the ICEBIO team, I contributed to and edited a “Frontiers for Young Minds” article on glacial habitats, aiming to communicate the science of these unique ecosystems to a younger audience.

DC5 ANIRBAN MAJUMDER january 25 - JUNE 25
Month 19 (Jan 25): The new year began with preparations for the winter fieldwork in Svalbard. Most of my time was dedicated to organising the logistics and planning for the expedition. Simultaneously, I was collaborating with colleagues from BAM on metabolomics bioinformatics data analysis and processing. I also submitted an abstract for EGU 2025.
Month 20 (Feb 25 ): was one of the members from the winter Svalbard fieldwork held between 8-22 February 2025. During this period, I collected ice cores and snow samples for Chapter 3 of my thesis.
Month 21 (March 25): This month, I primarily focused on metabolomics data analysis, particularly on understanding the pre-processing steps for mass spectrometry data.
Month 22 (April 25): Back in the lab, I resumed work on DNA/RNA co-extraction from samples collected during different field campaigns last year, 2024 and this year, 2025. At the same time, I continued with the data analysis of metabolomics datasets from both the Greenland fieldwork (Chapter 2) and the Svalbard fieldwork (Chapter 3). I also prepared a poster for the EGU 2025 conference.
Month 23 (May 25): I presented a poster at the European Geosciences Union (EGU 2025) conference, which was held in Vienna from 26 April to 2 May 2025. After the conference, I continued with lab work, focusing on DNA/RNA co-extraction. The extracted samples were then sent to our collaborators at RISO, Aarhus University, Denmark, for amplicon sequencing.
Month 24 (June 25): I continued the labwork with preparation for metabolite extraction. Also, the last set of DNA samples was sent to RISO for sequencing. Simultaneously, I was also doing metabolomics and amplicon 16S data analysis and structuring my thesis.
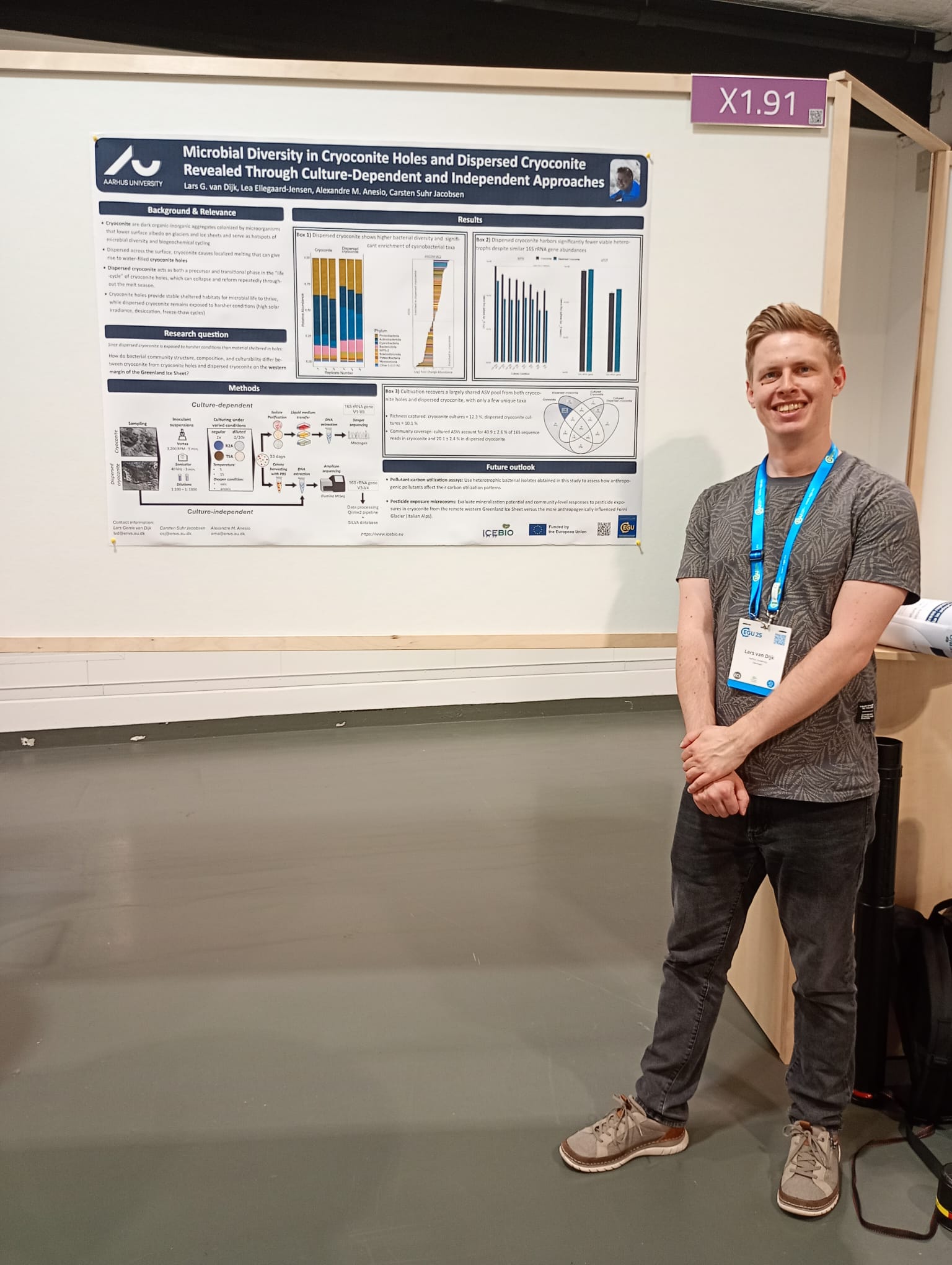
DC4 LARS VAN DIJK February 25 - July 25
Month 19 (Feb. 24): Regular sampling of the microcosms continued throughout the 170-day incubation experiment on pesticide degradation by cryoconite microbial communities (through the end of May). I organized department’s biannual PhD bundle meeting themed “Insights and Reflections on the PhD Experience” and attended the PhD writing camp, where I drafted my first manuscript, shared writing tips and tricks, and took part in small workshops.
Month 20: Following my January midterm examiner’s advice, I performed additional analyses on the microbial diversity of dispersed and submerged cryoconite. I also preread and gathered literature for the upcoming deliverable on the microbial diversity of the subglacial microbiome.
Month 21 (April 25): I presented my research via poster at the European Geosciences Union conference in Vienna and co-organized our department’s annual seminar on workplace inclusivity, centering on nationalities and cultural clashes.
Month 22:I ended the long-term incubation experiment on pesticide degradation by cryoconite microbial communities and prepared all samples taken during this period for DNA/RNA co-extraction. I also attempted to isolate single cyanobacterial filaments from my enrichments, which proved too challenging, so plate-streak isolation was done instead.
Month 23: Samples were selected and processed for DNA/RNA co-extraction. I then initiated qPCR on these extracts, targeting the 16S rRNA gene and key pesticide-degradation genes.
Month 24 (July 25): qPCR analyses continued, targeted isolation of pesticide-degrading microbes via enrichment plating was started, and samples were prepared and sent for total RNA metatranscriptomic sequencing.

DC9 KARA SAMPSELL: march 2025 - August 2025
Month 19: In March, I began working with the 16S rRNA gene sequencing data from the glacial flour samples and from our microcosm experiment and began planning exactly what to present at EGU in May. During this time, we also arranged for the last materials to be ready for the plant experiment to begin in May. We had several meetings regarding the plant experiment details and for planning secondment time at UiT. I did some gas measurements from the microcosm experiment with Roberto Grilli at IGE. I measured the particle size of remaining glacial flour samples at IGE as well.
In April, I finalized the contents of the presentation for EGU with further data analysis and work on the slides. We wrapped up planning the plant experiment and targeted a start date. From April 28th - May 2nd I attended the EGU conference.
In May, I began the plant experiment and went on field work to Chamonix to assist with Catherine’s ice core drilling mission. Upon our return, I began sampling for the plant experiment in later May. I also drafted a blog post for EGU Biogeosciences blog.
June consisted of more plant experiment sampling, sample processing, pH measurements, and DNA extractions. I worked a bit on continuing analysis of the microcosm experiment data. Silje, Marco, Klara, and I finalized one policy brief and wrote a second for IceBio.
In July, I continued the work for the plant experiment. Lars and I began working on our shared deliverable and convinced Léa to join us. I continued working on analysis of the microcosm experiment data and writing when possible.
Month 24: In August, I finalized details and prepared materials for the secondment. I wrote the second deliverable which was due in August. Both deliverables were submitted in the middle of the month. I concluded the plant experiment sampling mid-month as well. My second secondment began at UiT from the 18th of August. I wrote the report and made the presentation for my year 2 CSI meeting (institutional PhD progress/status check) which was completed on the 28th. I began extracting DNA from glacial sediments at UiT for the secondment.
DC2 HARPREET SINGH: March 2025 - august 2025
Month 19 (March 2025): We analysed and compiled the data on winter microbes of Arctic and organized the information based on 3 defined questions that explains about the active and total microbial structure and the overall metabolic processes related to microbial activity. We had another networking with Dr. James W Bradley group in Marseille where we exchanged our science and progress. Since I have a collaboration, we discussed more on our approach and possible direction of work.
April 2025: We prepared for another sampling session in Svalbard where we performed an in-situ microcosm experiment to mimic the event of rain on snow and how it impacts the microbial processes. Since winter in arctic are experiencing increase in precipitation, the rain could have a positive or negative effect on microbial activity and our experiment is to validate the response under such conditions. We completed the experiment and collected the materials for analyses.
May 2025: We attended the ICEBIO network meeting and the EGU25 conference in Vienna where I have presented my poster on the preliminary findings of microbial activity during winter. In parallel I was working on my article and mining literature for previous studies and findings.
June 2025: We initiated the preparation for 2nd and 3rd chapter of my thesis. We are targeting to compare our winter processes with spring as a chapter and the microcosm experiment to test the effect of rain on snow will act as another chapter of my thesis. Hence, we began preparing the samples for DNA and RNA extraction to perform metagenomic and totalRNA sequencing. Additionally, we have some chemical analyses to perform which will be continued in Aarhus University in Denmark during my secondment. Then I have another collaboration with the Italian team from Svalbard who will provide the inorganic analyses.
July 2025: We had another sampling campaign in Col du Dome, Mont blanc where we drilled the ice core for microbial ecology and it was a new exposure for us to work and understand the mechanistic of drilling at high altitude. It was a 2-week campaign where we learnt about the difficulties and management of such field trips. Nonetheless, we learnt about the properties of snow and ice of Col Du dome and its possible use in relating the past atmospheric composition with the signatures hidden in ice cores.
August 2025 (month 24) : We prepared the libraries of DNA and RNA and will finish the sequencing of both this year. Then we will begin the analyses for 2nd and 3rd Chapter. The last year of the PhD will be focussed on compiling the available information into publishable article and develop the knowledge on pre-existing gaps in these studies.
DC2 NASIM KASHIRI: May 2025 - Octobe 2025
Month 19 (May): After coming back from the EGU conference, I jumped back into lab work, continuing with the temperature tolerance and haemolytic activity tests. I prepared the extracted DNA from my samples and headed once again to Roskilde for the second part of my secondment. There, I ran nanopore sequencing on all my DNA samples. I also extracted DNA directly from my environmental samples and stored them there.
Most of June was spent sorting and processing all the sequencing data from the nanopore runs, which was a huge amount of data to take back with me! I also had an important meeting that slightly reshaped the plan for my first paper. After returning to Bremen, I took a short break for some vacation time.
Month 21 (July 2025): Back in Bremen, I started digging into the genomic data from Roskilde. I compared the genomes with pathogenicity- related databases to look for genes linked to virulence and antibiotic resistance. I also got everything ready for the antibiotic tests on the yeast isolates. By the end of the month, I was back in the lab to finish up some experiments related to my first manuscript.
Month 22 (August 2025): The first half of August was very lab-focused, finishing up all the experiments for the manuscript revisions and completing the antibiotic tests on the yeast isolates. I also prepared my first EU deliverables.
Months 23–24 (September–October 2025): These months were mostly dedicated to writing. I focused on wrapping up the first manuscript, going through feedback from my supervisors, and making revisions. It’s been busy but exciting to see the work coming together. By the last week of October, I was back in the lab again to run the antibiotic tests on all my isolates.

DC12 LEA FRANCOMME: MAY 2025 - october 2025
Month 19 (May): I started my secondment at Aarhus Universitet during which I performed the whole genome sequencing of Hydrurus foetidus via Nanopore sequencing and performed the assembly of the Hydrurus foetidus genome as well as initiated its annotation.
In June, I pursued my work on Hydrurus foetidus genome and extracted RNA from Hydrurus foetidus samples and prepared them for further sequencing using Illumina.
Month 21 (July): In July, I attended the FEMS 2025 conference in Milan during which I presented a poster about the microbial community associated to Hydrurus foetidus. I ran the last sequencing batches of the 16S RNA gene full length to complete the dataset about the microbial community associated with Hydrurus foetidus.
August was a short month due to vacations. Data analysis about the completed dataset for the Hydrurus foetidus associated microbial community was initiated.
In September, I attended the Cryospheric Ecosystem Conference in Poznan, Poland during which I gave a talk about the microbial community associated to Hydrurus foetidus. Global data preprocessing about the microbial community associated with Hydrurus foetidus was initiated as well as the preprocessing of the transcriptomic data produced earlier this year during my secondment at Aarhus University.
Month 24 (October): In October, preprocessing of both Hydrurus foetidus associated microbial community and Hydrurus foetidus transcriptomics datasets were achieved at 90%.











